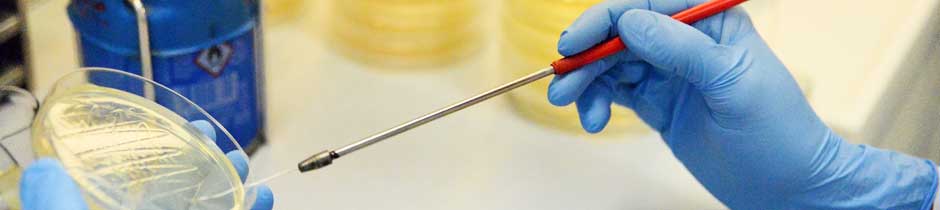
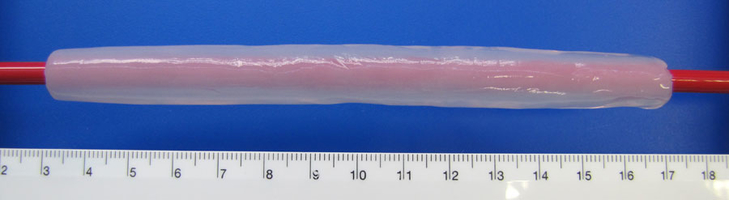
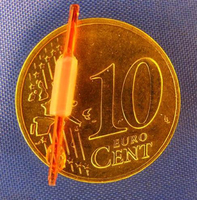
MMR-Tech-Tubes:
MMR-Tech enables the production of hydrogel bodies of any shape - for example tubular - directly during biotechnological synthesis. A key feature of this technology is that multilayer systems can be constructed, and additives can be incorporated directly into each layer during synthesis. The surface and network structures as well as the dimensions of the BNC tubes are also adjustable with this technology:
- variable inner diameter: 2.5-30 mm *
- Wall thickness: 1-5 mm *
- Length up to 175 mm *
- Bursting resistance: >1100 mbar internal pressure
Dry MMR-Tech tubes:
In post processing steps - e.g. partial or complete drying, laser structuring, sodium hydroxide shrinkage, etc. - the tubes can be further modified and their properties influenced.
- Inner diameter 5 mm
- Wall thickness < 100 µm
- Length 100 mm
BASYC-Tech-Tubes:
With the BASYC® technology, which is also patented, tubes with a particularly small inner diameter can be produced:
- Diameter inside 1 mm,
- Diameter outside 2.5 mm,
- length 10 mm *
* Dimensions: initial wet
Combining the outstanding properties of the natural polymer BNC with the process-controlled design of MMR-Tech, we obtain a wide range of tubes that are of particular interest for medical applications as implant materials for the replacement of hollow organs. We have years of experience in designing tubular implants for bile duct regeneration and blood vessel replacement.
Please contact us! We would be happy to work with you to develop new products.
Further literature:
BILE DUCT REGENERATION
Klemm, D.O.; Raddatz, V.;Klemm, D.O.; Raddatz, V.;Petzold-Welcke, K.; Kramer, F.; Ruhe,C.; Nietzsche, S.; Settmacher, U.;Rauchfuß, F. Preparation of TubularBiocellulose Implants and Its Use inSurgery—A Review. Processes 2021, 9,2114.
Klemm, D., Rauchfuss, F., Kramer, F., Petzold-Welcke, K., Richter, T., Ruhe, C., & Tannapfel, A. (2016). Design of biocellulose implants for first successful bile duct regeneration. American Chemical Society. 251st ACS National Meeting & Exposition, San Diego, United States.
Rauchfuss, F. (2018). Biocellulosis as scaffold for tissue regeneration in modern surgery. American Chemical Society. 225th ACS National Meeting & Exposition, New Orleans, United States.
Rauchfuss, F., Settmacher, U., Klemm, D., Fried, W., Richter, T., Petzold-Welcke, K., & Ruhe, C. (2017) WO2017021468A1.
CARDIOVASCULAR IMPLANTS
Klemm, D., Ahrem, H., Kramer, F., Fried, W., Wippermann, J., & Kinne, R. W. (2013). Bacterial nanocellulose hydrogels designed as bioartificial medical implants. In M. Gama, P. Gatenholm, & D. Klemm (Eds.), Bacterial nanocellulose: A sophisticated multifunctional material (pp. 175–196). Boca Raton: CRC Press.
Scherner, M., Reutter, S., Klemm, D., Sterner-Kock, A., Guschlbauer, M., Richter, T., Langebartels, G., Madershahian, N., Wahlers, T., & Wippermann, J. (2014). In vivo application of tissue-engineered blood vessels of bacterial cellulose as small arterial substitutes: Proof of concept? The Journal of Surgical Research, 189(2), 340–347.
Schumann, D. A., Wippermann, J., Klemm, D. O., Kramer, F., Koth, D., Kosmehl, H., Wahlers, T., & Salehi-Gelani, S. (2009). Artificial vascular implants from bacterial cellulose: Preliminary results of small arterial substitutes. Cellulose, 16(5), 877–885.
Wacker, M., Kießwetter, V., Slottosch, I., Awad, G., Paunel-Görgülü, A., Varghese, S., Klopfleisch, M., Kupitz, D., Klemm, D., Nietzsche, S., Petzold-Welcke, K., Kramer, F., Wippermann, J., Veluswamy, P., & Scherner, M. (2020). Hemo- and cytocompatibility of surface modified bacterial nanocellulose small diameter vascular grafts: an in vitro study. PLOS ONE.
Weber, C., Reinhardt, S., Eghbalzadeh, K., Wacker, M., Guschlbauer, M., Maul, A., Sterner- Kock, A., Wahlers, T., Wippermann, J., & Scherner, M. (2018). Patency and in vivo compatibility of bacterial nanocellulose grafts as small-diameter vascular substitute. Journal of Vascular Surgery, 68(6S), 177S-187S.e1.